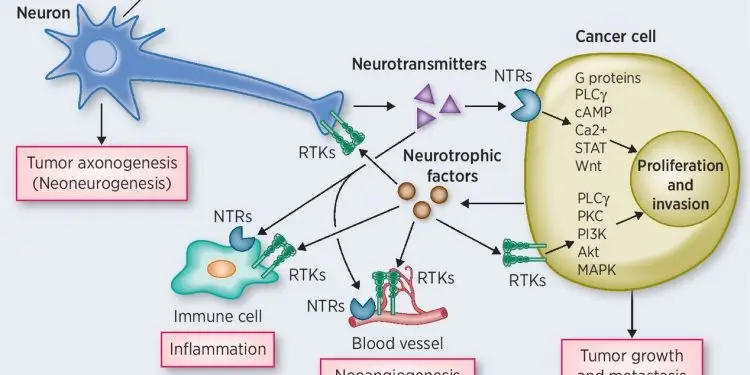
Blocking nerve growth factor receptor disrupted cholinergic signaling and prevented stomach cancer growth in mice
Gastric tumors are started by specialized cells in the stomach that signal nerves to make more acetylcholine, according to a study in mice. The multinational team of researchers who conducted the study also identified a substance called nerve growth factor that stimulates nerve development and, when blocked, inhibits stomach cancer development.
The findings were published today in Cancer Cell.
Previous studies have shown that nerves are abundant in the gastric tumor microenvironment. In an earlier paper, the researchers demonstrated that inhibiting signaling by the neurotransmitter acetylcholine, by severing the vagus nerve in the stomach or treating with Botulinum toxin, shrank or prevented the growth of gastric tumors in mouse models.
“Nerves and acetylcholine clearly play a key role in regulating the development and growth of cancer cells, particularly cancer stem cells, in the gastric tumor microenvironment,” said Timothy C. Wang, MD, the Dorothy L. and Daniel H. Silberberg Professor of Medicine at Columbia University Medical Center (CUMC) and senior author of the paper. “But little is known about what is driving cancer in the earliest stage of development, before the expansion of nerves in the microenvironment. We also wanted to find out where acetylcholine is coming from before the growth of nerves.”
Through a series of experiments in mouse models, the researchers determined that a neurotrophin (substance that triggers nerve growth) called nerve growth factor is highly expressed in gastric cancer cells. They also discovered that tuft cells–specialized cells found in the lining of the digestive tract that, like nerves, communicate with other cells–provide another source of acetylcholine for cancer cell growth, particularly during the formation of tumors.
“We learned that tuft cells are increased during the earliest stage of gastric tumor development, making acetylcholine and stimulating the production of nerve growth factor within the lining of the stomach,” said Dr. Wang. “As nerves grow in around the tumor, tuft cells decrease.”
In additional experiments, the scientists showed that overexpression of nerve growth factor in the mouse stomach drove tumorigenesis. Furthermore, administration of a nerve growth factor receptor inhibitor prevented stomach cancer in the mice.
“Our study provides some insight into the cellular crosstalk that leads to the development of stomach cancer, and points to a viable therapeutic target for this type of cancer,” said Dr. Wang. “Using our findings as a paradigm, additional studies can be done to identify the specific neurotrophins and neurotransmitters that are involved in tumor development in other areas of the body.”
###
The study is titled, “Nerve growth factor promotes gastric tumorigenesis through aberrant cholinergic signaling.” The other contributors are: Yoku Hayakawa (University of Tokyo, Tokyo, Japan), Kosuke Sakitani (University of Tokyo), Mitsuru Konishi (University of Tokyo), Samuel Asfaha (University of Western Ontario, Ontario, Canada), Ryota Niikura (University of Tokyo), Hiroyuki Tomita (Gifu University Graduate School of Medicine, Gifu, Japan), Bernhard W. Renz (Hospital of the University of Munich, Munich, Germany), Yagnesh Taylor (CUMC), Marina Macchini (CUMC). Moritz Middlehoff (CUMC), Zhengyu Jiang (CUMC), Takayuki Tenaka (CUMC), Zinaida A. Dubeykovskaya (CUMC), Woosook Kim (CUMC), Xiaowei Chen (CUMC), Aleksandra M. Urbanska (CUMC), Karan Nagar (CUMC), Christoph B. Westphalen (Klinikum der Universität München, Munich, Germany), Michael Quante (Technische Universität München, Munich, Germany), Chyuan-Sheng Lin (CUMC), Michael D. Gershon (CUMC), Akira Hara (Gifu University Graduate School of Medicine), Chun-Mei Zhao (Norwegian University of Science and Technology, Trondheim. Norway), Duan Chen (Norwegian University of Science and Technology), Daniel L. Worthley (University of Aidelaide, Australia), and Kazuhiko Koike (University of Tokyo).
The study was supported by grants from the National Institutes of Health (U54CA126513, R01CA093405, R01CA120979, and R01DK052778), the Clyde Wu Family Foundation, the Nakayama Cancer Research Institute, the Okinaka Memorial Institute for Medical Research, and the Project for Cancer Research and Therapeutic Evolution from the Japan Agency of Medical Research and Development. Y.H. and K.S. were supported by Japan Society for the Promotion of Science, and Y.H. and T.T. were supported by Uehara Memorial Foundation.
The authors declare no conflicts of interest.
Columbia University Medical Center provides international leadership in basic, preclinical, and clinical research; medical and health sciences education; and patient care. The medical center trains future leaders and includes the dedicated work of many physicians, scientists, public health professionals, dentists, and nurses at the College of Physicians and Surgeons, the Mailman School of Public Health, the College of Dental Medicine, the School of Nursing, the biomedical departments of the Graduate School of Arts and Sciences, and allied research centers and institutions. Columbia University Medical Center is home to the largest medical research enterprise in New York City and State and one of the largest faculty medical practices in the Northeast. The campus that Columbia University Medical Center shares with its hospital partner, NewYork-Presbyterian, is now called the Columbia University Irving Medical Center. For more information, visit cumc.columbia.edu or columbiadoctors.org.