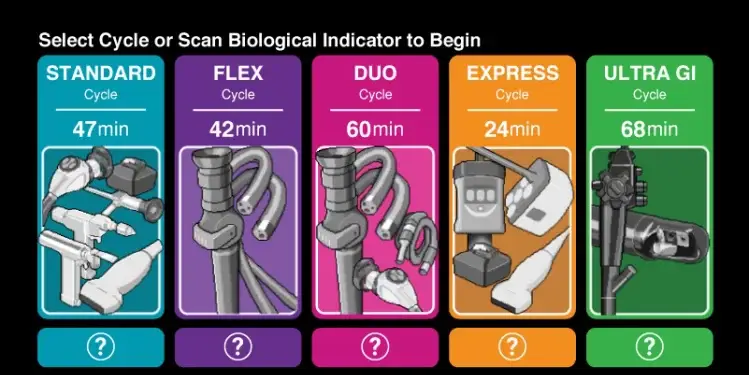
IRVINE, California: Advanced Sterilization Products (ASP), in collaboration with PENTAX Medical, announces the FDA clearance of the new ULTRA GI Cycle in its flagship product, the STERRAD 100NX Sterilizer with ALLClear Technology. The ULTRA GI Cycle is designed to reprocess duodenoscopes using hydrogen peroxide gas plasma sterilization.
The FDA clearance marks a significant milestone in advancing patient safety and addressing the challenges associated with current reprocessing methods for duodenoscopes. This sterilization cycle may revolutionize sterilization practices in healthcare facilities, according to a news release, ensuring a new standard of safety and efficacy.
Addressing Contamination Concerns
Duodenoscopes have been under scrutiny due to their relatively high contamination rates, prompting the urgent need for a robust sterilization solution. The collaborative effort between ASP and PENTAX Medical has resulted in an innovative method that not only meets but exceeds industry standards, providing healthcare professionals with a powerful tool to enhance patient safety.
“We’re proud and excited to receive FDA clearance for our new ULTRA GI™ Cycle, developed in partnership with PENTAX Medical,” said Chad Rohrer, President of ASP. “Both of our organizations are deeply committed to ensuring the safety of patients, and duodenoscopes have historically presented one of the greatest risks to patients in healthcare facilities. By using hydrogen peroxide gas plasma sterilization to reprocess duodenoscopes, we are achieving an increased level of certainty of sterility for healthcare professionals.”
“The use of hydrogen peroxide gas plasma represents a significant shift in making it the gold standard of sterilization for duodenoscopes and thereby improving safety for patients, technicians, and the environment,” said Ivan Salgo, MD, Chief Medical & Scientific Officer at ASP. “The ULTRA GI™ Cycle sets a new benchmark for the entire industry on sterilization and environmental safety and will give healthcare professionals and their patients a heightened level of assurance that the duodenoscopes used are now being sterilized in a safe and effective way.”
About Advanced Sterilization Products (ASP)
Advanced Sterilization Products (ASP) has a long track record of designing and delivering innovative infection prevention solutions that dramatically raise the level of health care and safety for those who matter most. Our pioneering technology, global distribution and established leadership position enable us to simplify the process of buying and operating infection prevention products and services every day for thousands of medical facilities around the world. This enables our customers to focus on what they do best – preventing infection and saving lives. For more information, please visit www.asp.com.